What are Geochemistry and Hydrochemistry?
Geochemistry is the study of the chemical composition of the materials found in the subsurface of the earth, and of the reactions that they undergo. In the context of geothermal engineering, we typically consider the geochemical and mineralogical composition of the reservoir rocks or sediments (primary geochemistry). We also consider the precipitates, scales and secondary minerals that may form in reservoir, wells or at the surface in heat exchangers and plant as a result of the operation of the geothermal scheme (secondary geochemistry).
Hydrochemistry is the study of the chemical composition of natural waters. Many deep geothermal fluids have rather unusual hydrochemical characteristics: they can be highly saline, very reducing or have high contents of dissolved gases (and the composition of these dissolved gases can be very important in many contexts). Also of interest is the composition of waters, acids and other fluids which are injected into the geothermal reservoir for purposes of hydraulic or chemical stimulation.
To successfully run a geothermal plant, the geothermal engineers need to understand the chemical interactions between fluids, rock minerals and gases during the stimulation and operation of a geothermal system. Therefore, the abrupt temperature changes that the fluids and rocks may undergo have to be taken into account. In particular, hydrochemists will consider how the compositions of the fluids, minerals and gases change throughout the lifetime of a geothermal site, in order to understand the chemical processes taking place in the reservoir. This will allow hydrochemists to evaluate whether these changes are likely to lead to the formation of secondary minerals, permeability decreases, scaling or clogging of the reservoir or of wells, or whether they may lead to permeability enhancement. This information must successfully be communicated to the geothermal engineers as clear recommendations for the stimulation or operation of the geothermal system.
Why is Hydrogeochemistry Important in the Context of Geothermal Operations?
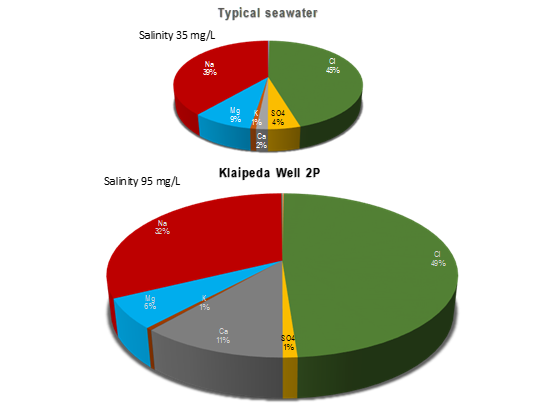
Geothermal boreholes are often very deep and provide a rare opportunity to glimpse some of the strange geochemical processes that occur at several kilometres depth in the earth’s crust. For example, the origin of the hypersaline brines (i.e. brines more concentrated than seawater) that are commonly found in the deep subsurface is still the subject of some academic debate. At the Klaipeda geothermal scheme in Lithuania, the groundwater has a dissolved solute content of some 95 grams per litre (g/L), compared with seawater at 35 g/L. Chemistry (and the isotopic composition of some of the chemical constituents) can be a useful tool in the academic understanding of the evolution of such brines (Figure 1).
Aside from the academic interest, geochemistry and hydrochemistry are of key importance for the successful operation of a geothermal scheme – because a poor understanding of chemistry can result in corrosion of plant, clogging of wells and reservoirs and may even cause environmental problems.
Clogging and corrosion
We all know that temperature affects solubility. Sugar dissolves more quickly in hot tea than in cold. Sugar can, on the other hand, start to crystallize out of boiling jam as it cools. The operation of a geothermal scheme typically leads to geothermal fluids being cooled. If we are not careful, the drop in temperature can lead to certain minerals (such as gypsum) precipitating out of the water and clogging up the heat exchangers, injection wells, pipes or the reservoir. Fortunately, geochemical software such as PHREEQC allows us to calculate the saturation index (a measure of whether a mineral tends to dissolve or precipitate) at different temperatures and to predict whether a problem is likely to occur (Figure 2). If a problem is predicted, remedial action can be taken: for example, at Klaipeda, the reinjected water is dosed with sodium phosphonate (DTPMP), which binds up the calcium in the water and helps to prevent gypsum precipitation.
Similarly, if a geothermal fluid contains a lot of dissolved iron or manganese, contact with atmospheric oxygen should be avoided. Otherwise the dissolved iron and manganese can oxidise and precipitate out as an orange or brown oxide / hydroxide sludge, which can quickly clog up heat exchangers and injection wells. Alternatively, hot, saline, reducing geothermal fluids can be very corrosive towards steel components (even some stainless steels) and a good knowledge of chemistry is necessary to predict the chemical risk.
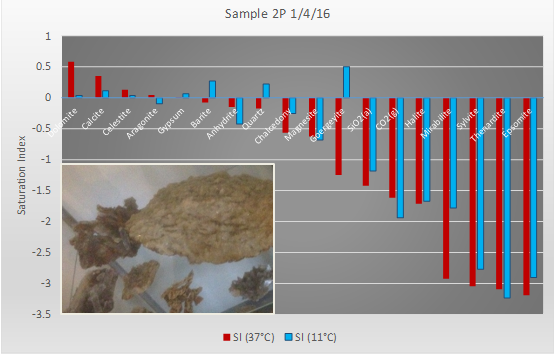
Figure 2. Saturation indices (SI) for water from geothermal well 2P at Klaipeda, Lithuania, calculated using the software PHREEQC at the production temperature (37°C, red) and the injection temperature (11°C, blue). Note that minerals such as dolomite, calcite and celestite have a positive SI at both temperatures and thus have a tendency to precipitate, although the SI is less at 11°C. Gypsum and barite, on the other hand, have SIs which become positive at 11°C and thus are more at risk of precipitating on cooling in the geothermal system. Minerals such as halite and mirabilite have negative SI at both temperatures and should not form precipitates. The inset shows fragments of gypsum that have precipitated out in the geothermal pipelines, exactly as predicted!
Acid stimulation
Acidisation is a technique widely used in both the water industry and the oil industry to increase the yield of wells. It is now also applied in the geothermal industry. The general idea is that a slug of acid is injected into the well. The acid dissolves minerals in the walls of fissures and fractures, enlarging their aperture and making them more permeable. Acid can also be used to dissolve scales in wells. However, not all minerals are soluble in all acids and the treatment must be tailored to the mineral assemblage in the rock (or scale) in question. For example, carbonate minerals are soluble in hydrochloric acid (among others) and this treatment has successfully been used for decades in many of the limestone and chalk wells from which we obtain our drinking water. However, most silicate minerals are not susceptible to attack by hydrochloric acid. Indeed, to make an impact on silicate formations, hydrofluoric acid has been used.
Environmental concerns
When drilling into deep geothermal reservoirs, it is important to realise that the (completely natural) fluids encountered at depth may contain concentrations of elements or chemicals that should not be discharged unmanaged to the surface environment. When geothermal boreholes are artificially hydraulically stimulated, relatively benign surface water may be injected into the well. When the stimulation is completed, however, the “flowback” water emerging from the well may contain a significant component of deep formation water. Figure 3 shows the evolving composition of the flowback water following geothermal well stimulation in a granite aquifer at Pohang, South Korea.
The natural chemicals contained in deep geothermal formation water may range from hydrogen sulphide gas (which pongs of rotten eggs and may be unpleasant for neighbouring landowners), or salinity, to low concentrations of the metal barium or naturally occurring radioactive materials (NORMs) such as radium. Indeed, such NORMs may form solid scales on the walls of some hydrocarbon and geothermal boreholes or pipelines, and may need to be carefully managed when removed via descaling activities. If the presence of these natural chemicals is recognised, transparently discussed and understood, appropriate management and disposal pathways can be identified – as indeed has been the case in the hydrocarbon industries for many years.
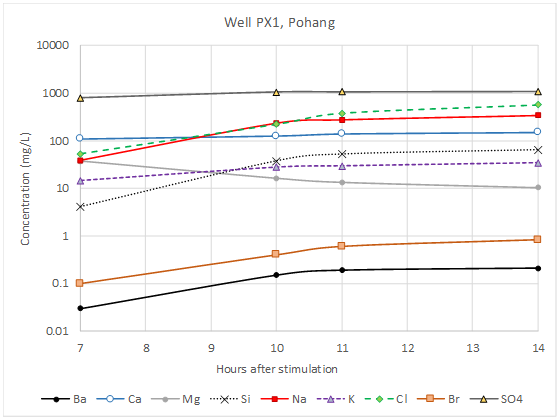
Figure 3. Change in concentrations of key hydrochemical parameters in flowback water following hydraulic stimulation of Well PX-1 at Pohang, Korea on 23rd January 2017. Note the increase in sodium, chloride, silicon and barium; all these parameters are characteristic of a deep, warm, reducing granitic environment. Reproduced by kind permission of Prof. Eunhyea Chung of Seoul National University.
How to Design a Sampling and Analysis Program for a Geothermal Project
This is a huge and complex topic and only barest essentials will be touched upon here.
It is important to define the objective of a sampling campaign. A focus is required on what exactly needs to be sampled, and when, and also for which parameters the samples should be analysed. Practical procedures for water sampling are described in the United States Geological Survey online Field Guide (and by various national and European standards). Particular attention should be paid to the practice of field filtration of water samples (such that particles and drilling cuttings do not interfere with analyses) and appropriate sample preservation.
It is always beneficial to obtain samples of the geothermal reservoir rock, either by coring of an on-site borehole, or by obtaining cores from an off-site borehole into the same geological stratum. These can be supplemented by samples of disturbed drilling cuttings or samples of the reservoir rock from outcrops in the vicinity (if any). Rock / sediment samples can be analysed by:
- X-Ray diffraction techniques to ascertain which minerals are present.
- X-Ray fluorescence, and other techniques, to ascertain the elemental composition of the rock.
- Water or acid digestion trials to ascertain which elements are readily released to water upon contact and hydrolysis.
- Where acid stimulation is proposed, real samples of the rock can be subject to acid exposure under controlled laboratory conditions to study the effects.
As regards water sampling, samples should be taken at or near the well head (subject, of course, to safety considerations) and determinations of the following characteristics should be made in the field: temperature, electrical conductivity, dissolved oxygen, redox potential, pH, alkalinity, CO2 content and redox-sensitive parameters such as hydrogen sulphide.
Easily operated hand-held meters or kits are available to measure many of these parameters on-site. Fluid samples can be analysed in the laboratory in order to gain information on:
- Fluid composition, by determining major and minor ion contents or gas components
- The age, origin and history of the water by measuring isotopes
Samples of geothermal water should always be analysed (in the laboratory) for concentrations of major ions, namely: sodium, calcium, potassium, magnesium, ammonium, chloride, carbonate/bicarbonate, sulphate and nitrate.
In addition, the following parameters will usually prove invaluable in interpreting the water chemistry and assessing its suitability for geothermal exchange: strontium, barium, lithium, boron, bromide, fluoride, silicon, iron, manganese, arsenic, sulphide, uranium, total dissolved solids, total suspended solids, stable isotopes of oxygen (18O), hydrogen (2H), sulphur (34S) and carbon (13C). Natural radioactive isotopes, such as 14C, can sometimes be useful in estimating the age of the water.
Often, in addition to analysing water from deep geothermal wells, it is common to carry out a campaign to collect water samples from other nearby springs and wells. Comparing the hydrochemistry may help us to understand any hydraulic connection between a deep geothermal reservoir and the shallow groundwater system. Any warm springs near a geothermal operation can be a mixture of deep geothermal fluids and shallow groundwater; - hydrochemistry can assist us in estimating the extent of mixing and the degree of cooling during its journey to the surface.
Further reading
“Acidisation: borehole development and rehabilitation” , in Quarterly Journal of Engineering Geology by Banks et al. (1993). This review presents the use of acid as a technique for permeability enhancement and well yield stimulation in the public water supply industry in the latter part of the 20th Century, with emphasis on the Cretaceous Chalk aquifer.
“Acidizing Treatment in Oil and Gas Operators“, factsheet produced by the American Petroleum Institute (2014). This describes the use of various acid treatments, including hydraulic stimulation with acids, in the oil and gas industries.
“Review on chemical stimulation techniques in oil industry and applications to geothermal system”, a CREGE report by Portier et al. (2007, and
“Chemical stimulation techniques for geothermal wells: experiments on the three-well EGS system at Soultz-sous-Forêts, France”, in Geothermics by Portier et al. (2009). These papers review and describe the use of various chemical stimulation techniques at real geothermal sites.
“Using Kr and noble gases to characterize and date groundwater and brines in the Baltic Artesian Basin on the one-million-year timescale”, in Geochimica et Cosmochimica Acta, by Gerber et al. (2017). This research paper describes how groundwater chemistry and isotopic data can be used to understand the evolution and possible age of brines in a geothermal reservoir rock: in this case, the Devonian sandstone reservoir exploited by the Klaipeda (Lithuania) geothermal plant.
“Naturally Occurring Radioactive Materials (NORM) in Produced Water and Oil-Field Equipment - An Issue for the Energy Industry”, a factsheet produced by the US Geological Survey (1999). This factsheet describes the occurrence of NORM in deep geological fluids, focussing especially on hydrocarbon production.
“Chemistry of Thermal Fluids” represents Chapter 4 of an e-learning platform developed by the EU-funded GeoCom (Geothermal Communities) project. It provides an introduction to the thermal properties of geothermal fluids and to best practice in managing them.
“National Field Manual for the Collection of Water-Quality Data (NFM)” is kept constantly updated by the US Geological Survey. It provides up-to-date best practice for sampling natural waters and is broadly applicable to geothermal fluids.